A Quality Management System is a structured collection of integrated policies, processes, and procedures that bring all these elements into one system to achieve remarkable business success. The main functional elements of a QMS solution are quality policy, procedures, and work instructions. You can get benefits by using an automated QMS system to simplify, clarify, and control your manual process. Our QISS QMS system is the ultimate solution for organizations to achieve their quality vision, mission, goal, and objective. Everything in an organization needs to be documented, but it doesn't have to be paper documents. It is easier to store and maintain them in an electronic format using a Quality Management System.
Our QISS QMS system is designed to simplify your organization's quality policy. It helps you process clear work instructions for your organization, reducing rework and defects. Implementing QMS allows you to control your process more effectively and efficiently.
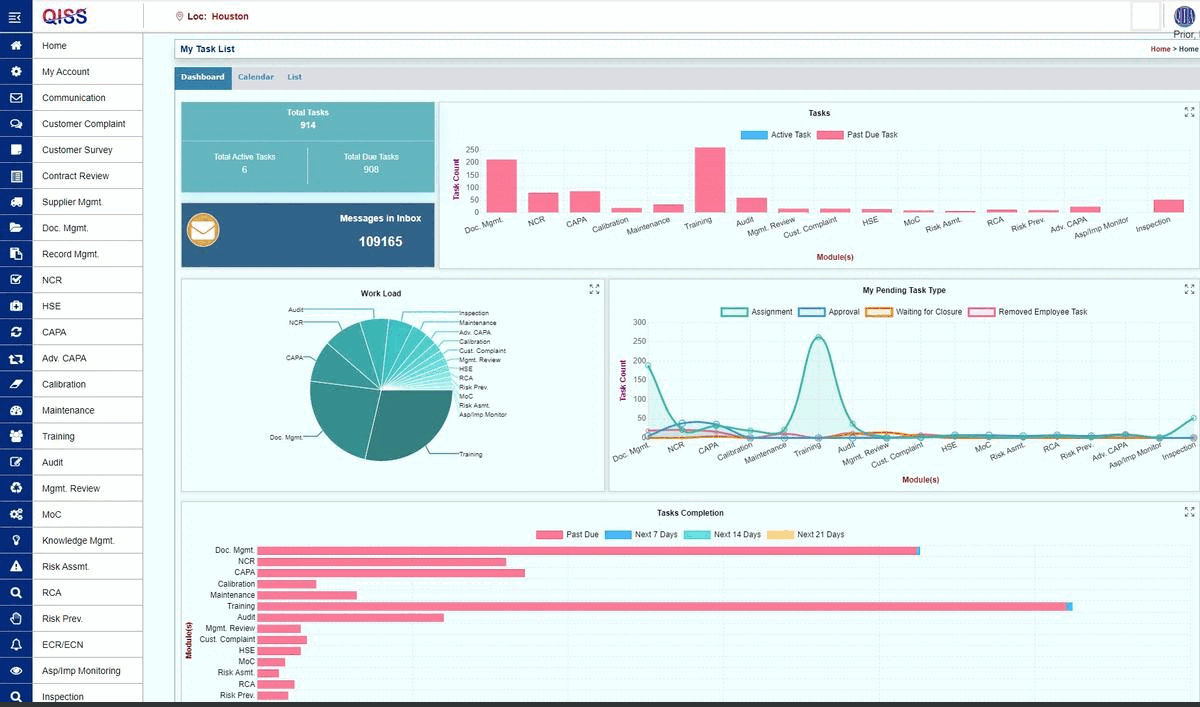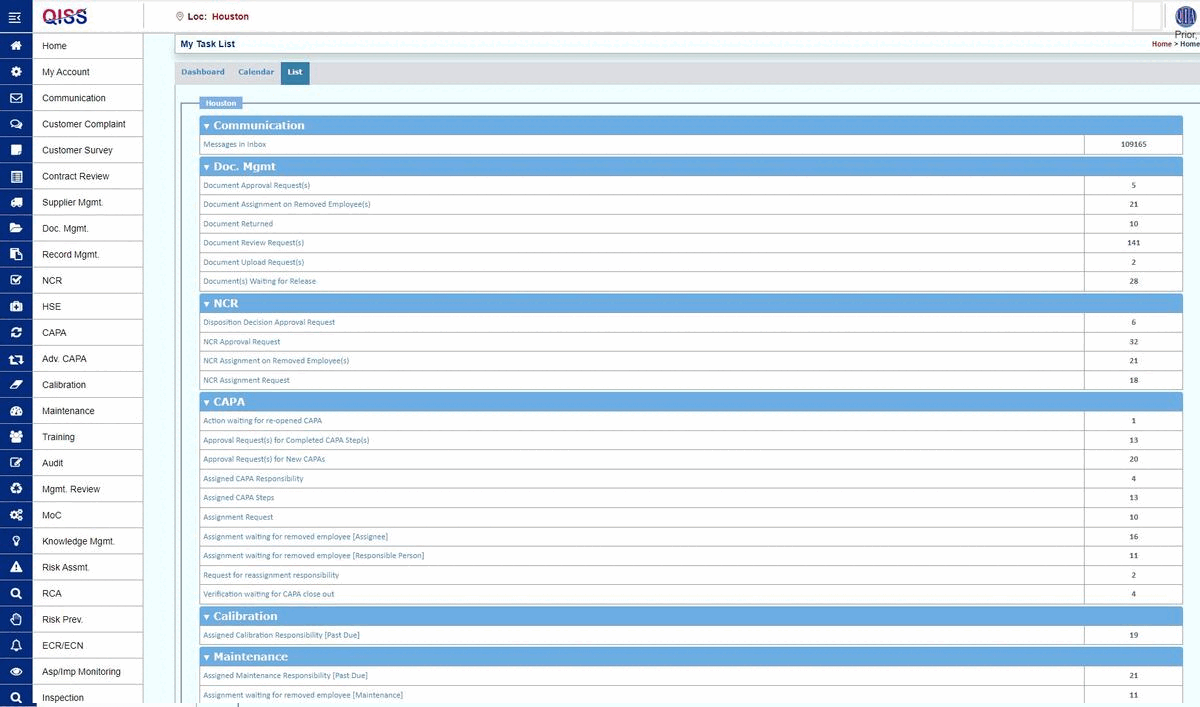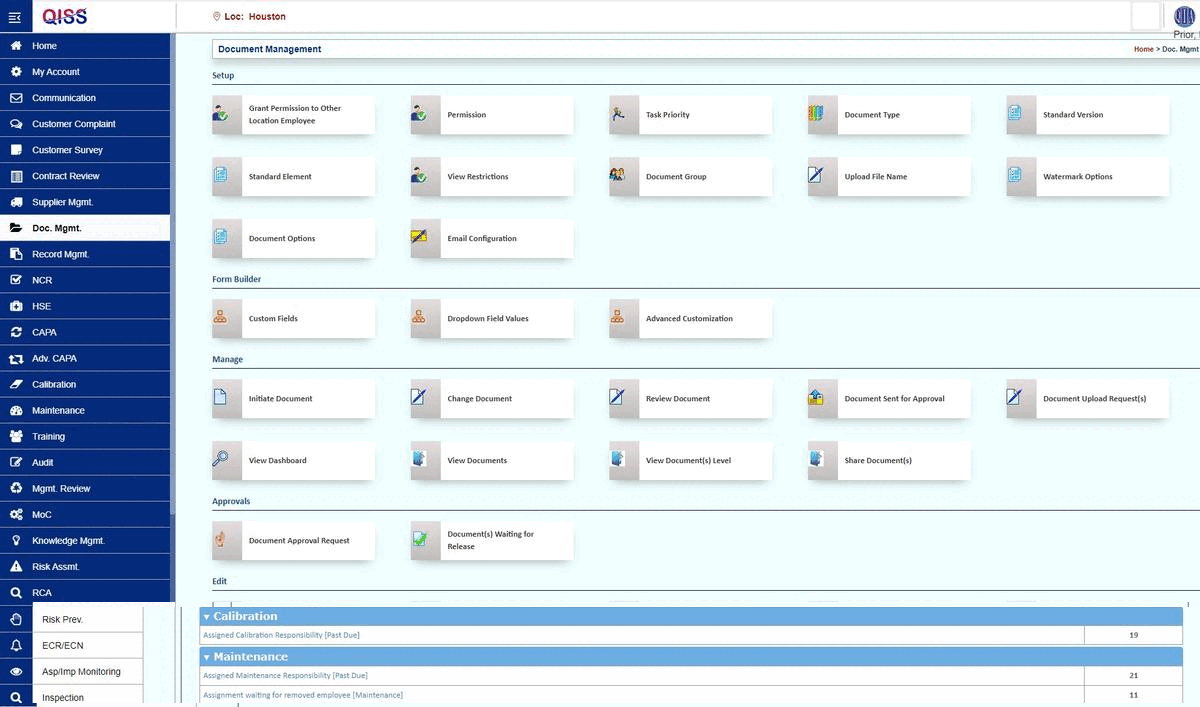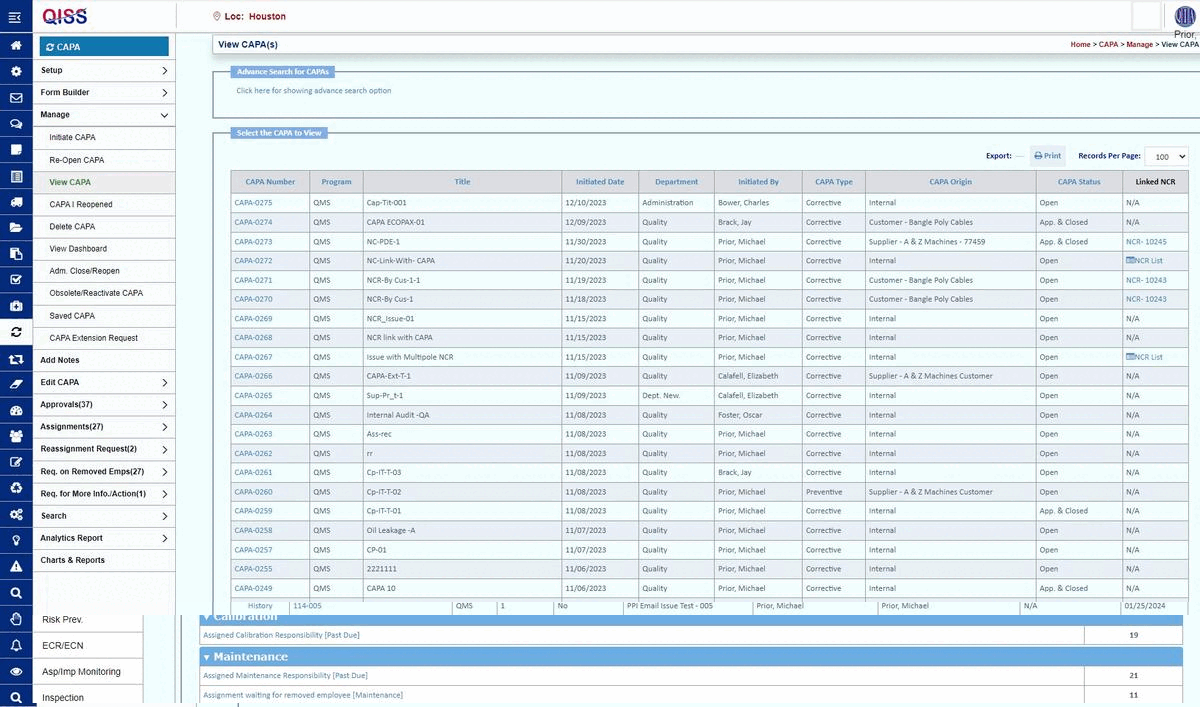
QISS was developed first for automating Quality Management Systems (QMS). The initial standard supported was ISO 9001, arguably the base standard for all subsequent standards.
What is Quality Management System (QMS)?
Quality management system is the process and procedure of an organization to deliver quality products and services. The main purpose of a QMS is to improve the overall efficiency of a business, reduce waste and errors, and increase customer satisfaction. It involves establishing a clear set of standards and procedures for all aspects of the company, from product design to delivery. A quality management system is built on several key principles. Customer focus, employee involvement and continuous improvement are the main elements of a well implemented QMS.
What does QMS Stand for?
QMS is the abbreviation for Quality Management System. There are several different QMS standards, including ISO/TS 16949, ISO 9001, ISO 14000, ISO 13485, and ISO 19011. Each of these standards provides a different framework, but they all share the same goal of improving the quality of products and services. It can help your company to improve your overall performance and stay competitive in today's market. Our QISS QMS solution helps you to increase customer satisfaction, reduce waste and errors, and improve your bottom line.
What is ISO 9001 Quality Management System?
The most popular quality management standard worldwide is ISO 9001. It has been issued to organizations in 189 countries and has over a million certificates. The only standard that can be certified within the ISO 9000 family is ISO 9001. It defines seven quality management principles, such as a strong customer focus and continual improvement.
Businesses Benefit from ISO 9001 in several ways:
Client Satisfaction: The standard guarantees that businesses have strong quality assurance procedures in place, this increases client confidence.
Efficient Handling of Customer Complaints: ISO 9001 provides guidelines for handling customer complaints effectively; this helps to solve problems in a timely and satisfactory manner.
Process Improvement: By identifying and removing inefficiencies, the standard lowers costs and produces better results.
Continuous Optimization: Consistent audits and evaluations, promoted by ISO 9001, help companies improve their quality management systems over time. This helps them maintain their competitive edge and success in the long run.
Why Implement a QMS?
Quality assurance and quality control are the two main components of a QMS system. To run an organization in a systematic way, you need to implement a QMS system. By implementing a QMS, an organization can fulfill customer requirements and legal compliance. Well-implemented quality assurance and quality control help you increase your business capability.
You need to follow five standards to implement a QMS system.
- ISO 9001 For Implementing QMS in all types of Industry
- ISO 14001 For Implementing EMS (Environmental Management System) in all type of Industry.
- ISO 45001 For Implementing OHSAS (Occupational Health and Safety Assessment Series) in all types of Industries.
- IATF 16949 (International Automotive Task Force) For implementing QMS in Automotive Industries.
- ISO 27001 Information Security Management System
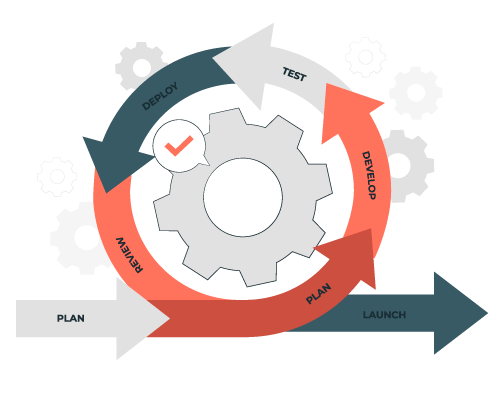
Quality Management Process
The quality management process is a series of procedures. It's used to verify that a team's deliverables are fit for purpose. This procedure is important in every business. It ensures that the deliverables provided truly fulfill the consumer's needs. The quality management process will assist you in immediately improving the quality of your deliveries.
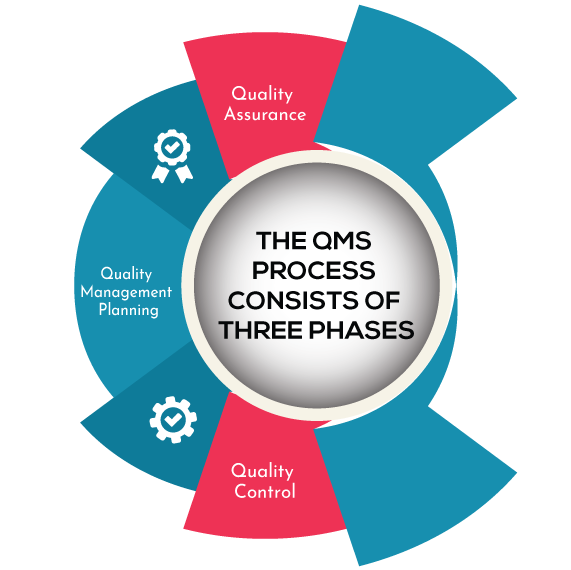
The QMS process consists of three phases: planning, assurance, and control. It ensures that deliverables meet consumer needs and improve quality.
The QMS process consists of three phases: planning, assurance, and control. It ensures that deliverables meet consumer needs and improve quality.
Quality Management Planing: Quality planning is the first phase. Planning of the quality process is explained
- Enterprise Environmental Factors
- Organizational Process assets
- Project Scope Statement
- Project Management plan
We have organizational process assets and work performance initiation followed by quality metrics and quality checklists.
Quality Assurance:
- Quality Management plan
- Quality Metrics
- Quality Process Improvements
- Performance Information
- Approved Change Requests
- Quality Controlled Measurement
Quality Control:
- Quality Management plan
- Quality Metrics
- Organizational Process assets
- Work Performance Informantion
- Approved Change Requests
Why is Quality Management Required?
Enterprises around the world are prioritizing themselves to provide quality goods. Today’s customers have a huge number of options in front of them only if the organization has quality goods. Enhancing quality through continuous improvements is such a vital role for many organizations. Nowadays, it is essential for an organization to have quality management to grab the customer's attention.
Why Quality Management System (QMS) is important to an organization?
Now, let us point to the checklist of why quality management is important.

- Creates Customer Satisfaction
Customer satisfaction is crucial for businesses to achieve loyalty and returning repeat customers, positive reviews, and a positive brand image. - Product consistency in terms of quality and design
Firms must plan, create, execute, and manufacture to ensure product consistency and accuracy. - Ensures long-lasting efficiency
Enhancing your QMS to achieve even greater efficiency and excellence. Quality Management is crucial for enhancing efficiency and product quality in any organization. It ensures that all employees, from engineers to sales managers, are aware of the best practices in their respective fields. - Reduces risks.
By lowering the risk, customers can be confident in receiving expected products or services. - Increases profits.
Enhancing product quality, reducing costs, and establishing a reputation significantly increase your revenue. - Maintains the competitive advantage.
Maintaining a high level of customer experience will sustain our competitive advantage.
Optimize your quality control and compliance with our QMS system. QISS QMS, is a comprehensive quality management software to achieve your goal. And discover the advantages of streamlined operations and improved regulatory conformance.
Book a personalized demo to learn more an Automated Quality Management System.
QISS QMS offers quality management software for all small and large companies, designed for regulatory compliance. QISS QMS is the all-in-one solution that is compliant, audit-ready, and user-friendly.
What are the benefits of a Quality Management System?
Effective use of Quality Management Systems (QMS) provides numerous benefits to organizations, including standardization of processes. And it also reduces the number of errors, and consistent delivery quality products or services. A quality management system is a tool that ensures conformity to your organization for quality standards and regulations. QISS QMS have the all capability to meet this requirement that benefits you.
- Compliance with quality standards: A QMS system ensures the conformity to the quality standards and regulations that are applicable to your organization.
- Improved Quality: A Quality Management System (QMS) improves quality processes and promotes ongoing improvement. This boosts the overall quality of your products or services.
- Improved Customer Satisfaction: Customer satisfaction is a key factor in retaining business. A QMS helps meet customer needs and address their concerns.
- Enhanced Efficiency: This system improved efficiency and productivity by eliminating waste and streamlining processes.
- Reduced costs: QMS system can significantly minimize the costs by enhancing quality and ensuring compliance, customer complaints and rework.
- Improved communication and collaboration: A quality management system involving employees and giving them clear objectives among various departments and teams.
- Your Companies can enhance profitability in 2024 and beyond by using these QMS benefits. This way you can position yourself as more competitive, original, and aligned with market needs.
What is the purpose of an Automated Quality Management System?
An Automated Quality Management System QMS collects and analyzes data, informing leadership and improving decision-making. QMS encourages proactive process enhancement, continuously improving operations. An Automated QMS identifies potential issues before they become problems, helping organizations manage and mitigate the risks. A well-implemented QMS aligns quality objectives with the overall business strategy, providing a competitive advantage. By integrate a fully automated QMS solution, like QISS QMS, you will also be able to save your valuable time and money.
Book a personalized demo with our teams to explore our QISS QMS Software.
Why Implement a QMS?
In 2024, implementing a QMS System can significantly enhance profitability for companies through strategic improvements and optimizations.
Process Optimization: QMS can streamline processes, reduce redundancy, and eliminate wasteful practices.
Enhanced Product Quality: QMS standardizes processes, adding feedback loops for high-quality outputs, reducing returns and rework, and increasing customer satisfaction.
Regulatory Compliance: QMS ensures industry regulations are followed, preventing costly penalties, fines, and reputational damage.
Data-Driven Decisions: A QMS system implementation always enables data collection, analysis and enabling informed decision making.
Employee Engagement: A Quality Management System (QMS) can boost employee productivity and profitability by involving them in quality initiatives.
Risk Management: A QMS is a tool that proactively identifies and manages risks, thereby reducing potential losses and ensuring business continuity.
Supply Chain Management: A Quality Management System (QMS) can improve quality control by ensuring reliable supply chains, reducing delays, and improving product delivery timelines.
Our software includes pre-configured workflows for document control, change control, non-conformance management, and CAPA management. Implementing a QISS QMS solution saves your time and money on quality system validation and continuous re-validation.
How can Companies Improve Profitability using Quality Management System (QMS) in 2024?
Fulfilling customer needs with quality products and services will increase revenue and repeat business. Commitment to quality helps expand market share and increasing customer loyalty. QMS creates an environment for continuous improvement that can improve products, services, and processes. These innovative solutions drive profits in your organization on a consistent basis. Implementing a Quality Management System (QMS) can boost sustainable business practices. And these are crucial for consumers, long-term customer relationships, and profitability.
How to implement iso 9001 quality management system?
◾Element and Requirements of a QMS
A QMS system consists of various components that work together to meet both customer and organizational requirements. ISO 9001 Quality management systems should sustain an organization's specific requirements, but some of elements are common in all system such as:
The organization’s quality policy and quality objectives
Quality manual
Procedures, instructions, and records
Data management
Internal processes
Customer satisfaction from product quality
Improvement opportunities
Quality analysis
◾Functions of a QMS
A quality management system should contain tools, processes, and resources to enable your company to complete its ISO 9001 QMS standards. These are the following key functions of a QMS:
Document Control: Ensure to use current documents and review and approve the changes. Keep the document records for the required duration.
Corrective and Preventive Action (CAPA) Management: The focus is on identifying, resolving, and preventing product compliance issues.
Training Control: We ensure that all staff training is regularly updated and recorded.
Audit Management: Quality audits are capable of ensuring the Quality Management System's (QMS) effectiveness in producing safe and compliant products.
Risk Management: The role involves proactive quality issue identification and control. And involving new product development, design changes, manufacturing, CAPA, and postmarket oversight.
What are the Principal elements of a Quality Management System?
ISO 9001 is based on 7 quality management principles. These are defined in ISO 9000:2015 and ISO 9004:2009. They are intended to provide senior management with a framework for improving performance within the organization. Let's look at each one of those principles right now.
Customer Focus: The first principle is customer focus in quality management. It maintains that organizations should understand current and future customer needs and requirements.
Leadership: The second principle is leadership. We all know that strong leadership is key to the success of any business. Leaders in an organization set the direction and create an environment for people to buy in and get involved.
Engagement of People: The next quality management principle is engagement of people. Leaders set the tone, but the benefits are truly apparent when you have full involvement of the people at all levels of the organization. This involvement promotes innovation, creativity, and accountability among other benefits.
Process Approach: The fourth principle is a process approach. By managing resources and activities as a process, a desired outcome can be obtained far more efficiently and effectively. This will improve costs and achieve predictable and consistent results.
Improvement: This is highly overlooked by many organizations. However, improvement should be a high-focus area and a constant objective for all levels of organizational performance.
Evidence-based Decision-making: The 6th quality management principle is evidence-based decision-making. It promotes making informed, fact-based decisions through careful analysis.
Relationship Management: The final principle is relationship management in quality management. The organization and its external partners must have an interdependent relationship that promotes value. They must work together to achieve consistent results that create value for both parties.
These are the 7 principles for quality management that structure the ISO 9001:2015 standard. It is wise for an organization to build off these principles when developing a quality management system. QISS QMS is the all-in-one solution that fulfill all the auditable requirements.
Book a personalized demo with our teams to explore our QISS QMS Software.
Benefits of QISS Quality Management System
- Assists in meeting Quality, Health, Safety, and Environmental (QHSE) standards such as those set by ISO.
- Extends beyond regulatory requirements to reap financial ($) rewards from an efficiently run business by means of well-designed, automated processes for ensuring compliance and overseeing management.
- QISS allows for granular control over who can do what within the system by modifying access levels.
- Capability in multiple regions and locations. In addition, areas within regions can be grouped together if desired.
- A location can either operate independently or in tandem with other locations.
- Capability to manage multiple programs at once (including quality, safety, and environmental). Various establishments can use their own unique software.
- Permissions determine what a user can and cannot do in each module. Each program's module manager or higher authority (location, regional, and QISS administrator) must grant access to that program's module before any action can be taken within it.
- Editing and viewing privileges can be set on a global, regional, or local level.
- Connecting users in other locations to what's happening locally.
- A queue of pending tasks that the user must complete.
- Email reminders of upcoming and overdue tasks, as well as general information.
- Exporting search results to Excel for additional analysis and report building.
QISS Key Features:
- Ease of Use: Intuitive interface for all users.
- Scalability: Grow with QISS as your needs evolve.
- Transparency: Crystal-clear data reporting.
- Integration: Seamlessly integrate with modern technology.
- Teamwork: Promote collaboration across your organization.
- Risk Control: Mitigate risks effectively.
- Whole-Quality Solution: Cover all aspects of QHSE.
- Advanced Analytics: Enhance insights with QISS. Create custom queries and generate reports for quality management.
QMS is an over-arching methodology for setting up and operating a system of governance for organizations, particularly those that produce a product for sale. The aim is to enhance Quality, Productivity and Profitability. This can be done in a manual fashion and can be done with the help of automation. In 2003, the Quality Institute of America (QIA) launched the Quality Institute Software Solution (QISS) to provide automation for the QMS. QISS has grown now to thirty independent but integrated modules. Organizations of all types and sizes can use one or all modules, as it suits them. The best way to learn about QISS is to request a live demo.
